BaoBab - ein Helmholtz-Innovationslabor
Eine Plattform zur umfassenden Isolierung und Charakterisierung humaner monoklonaler Antikörper
BaoBab ("Brain antibody-omics and B-cell Lab", Gehirn-Antikörperomik und B-Zell-Labor), wurde 2020 als Helmholtz Innovation Lab (HIL) gegründet und ist dem DZNE an den Standorten Berlin und Bonn angegliedert. Das wesentliche Element von BaoBab bildet eine maßgeschneiderte halbautomatische Plattform zur Generierung und Charakterisierung humaner monoklonaler Antikörper. Schwerpunkt ist die Isolierung von autoreaktiven Antikörpern aus Liquor- und Blutproben von Patienten. Damit wollen wir ein besseres Verständnis für die Rolle des humoralen Immunsystems bei einem breiten Spektrum neuropsychiatrischer und neurodegenerativer Erkrankungen erreichen. BaoBab ist strategisch darauf ausgerichtet, durch neue Einblicke in die Krankheitspathologie die Zusammenarbeit zwischen industriellen und akademischen Partnern mit dem DZNE zu fördern.
Technologien
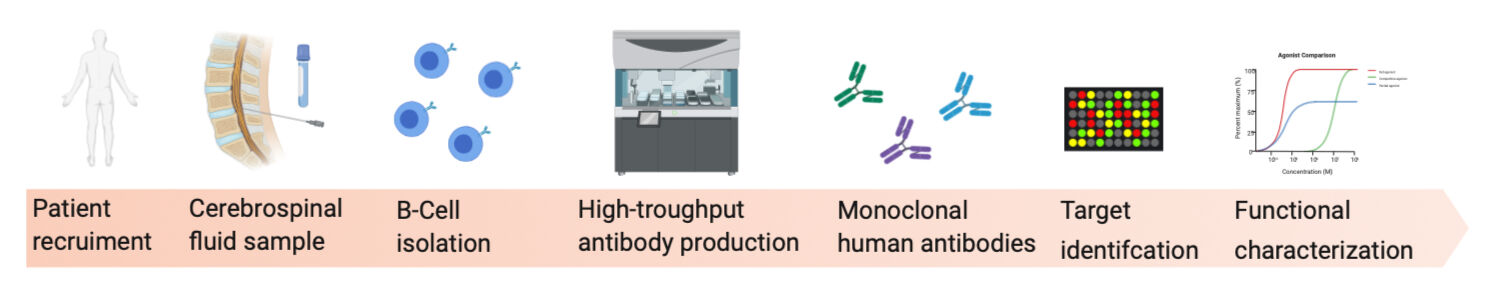
Humane Antikörper aus dem Liquor sind eine einzigartige Quelle bioaktiver Moleküle mit einem enormen akademischen und kommerziellen Potenzial. BaoBab hat es sich zur Aufgabe gemacht, dieses Potenzial zu erschließen, indem es neurologisch relevante Kohorten mit der Autoantikörper-Expertise der Arbeitsgruppe von Prof. Prüß in Berlin und der technischen Expertise der DZNE Core Research Facilities and Services (CRFS) unter Leitung von Dr. Eugenio Fava in Bonn verknüpft.
Die Arbeitsgruppe "Autoimmune Enzephalopathien" von Prof. Harald Prüß am DZNE Berlin verbindet klinische Tätigkeiten und translationale Forschung zu Antikörper-vermittelten Erkrankungen des Gehirns, wie Enzephalitis und Autoimmun-Demenz. Wir haben eine kleinmaßstäbliche Pipeline zur Isolierung und anschließenden rekombinanten Produktion von monoklonalen humanen Antikörpern etabliert. Im Rahmen unserer ersten Studien haben wir bereits mehrere hochgradig krankheitsrelevante humane Autoantikörper gegen eine Vielzahl von Zielmolekülen identifiziert und charakterisiert, darunter NMDA- oder GABA-Rezeptoren, LGI 1, Caspr2, GFAP, mGluR5 und den Acetylcholinrezeptor. Wir charakterisieren diese Antikörper durch eine breite Palette von experimentellen Ansätzen, sowohl in vitro als auch in vivo. Dies hat zu wachsender Expertise in einem breiten Spektrum von Technologien geführt, von der Immunfluoreszenz-Bildgebung bis zu Verhaltenstests an Nagetiermodellen.
Die Expertise der Core Research Facility reicht von der Lichtmikroskopie über die Bild- und Datenanalyse bis hin zu Laborautomatisierungstechnologien und wird durch ein Biorepository ergänzt. Modernste Automatisierungstechnologien ermöglichen es uns, eine Hochdurchsatz-Pipeline für die Generierung von monoklonalen Antikörpern zu etablieren. Wir haben eine dedizierte Plattform um eine zentrale Pipettiereinheit (i7, Beckman Coulter) herum entwickelt, die alle notwendigen Arbeitsschritte in den Bereichen Molekularbiologie (PCR-Maschinen: ATC - ThermoFischer, RT-PCR: LightCycler 480 - Roche, Reagenzienspotting: Echo 525 - Labcyte, Kapillarelektrophorese: Fragment Analyzer - Agilent, Spektroskopie: Big Lunatic - Unchained Labs), Mikrobiologie (QPix 460, Molecular Devices) und Zellkulturen ausführt. Um unsere Aktivitäten zu erweitern, verfügen wir darüber hinaus über weitere Plattformen und Instrumente wie ein konfokales Hochdurchsatz-Screening-Mikroskop (CV6000, Yokogawa).
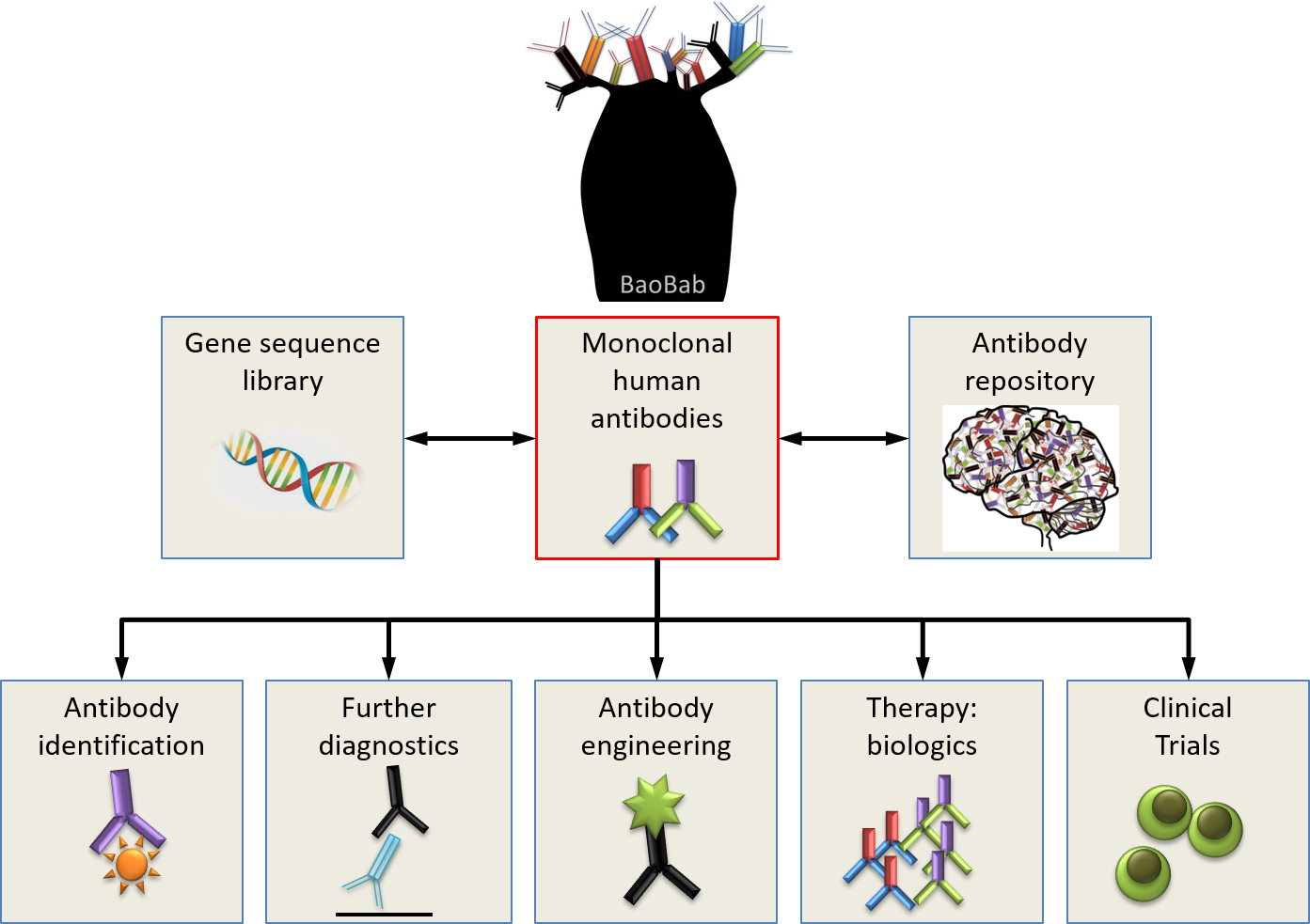
Unsere Antikörperdatenbank wächst ständig und wird für medizinische Forschungszwecke von großem Nutzen sein. Darüber hinaus wird sie die Entwicklung neuartiger diagnostischer Verfahren und innovativer therapeutischer Ansätze sowie wichtige Erkenntnisse über die Immunbiologie des zentralen Nervensystems ermöglichen.
Produkte und Partnerschaften
BaoBab bietet Zugang zu einer weltweit einzigartigen Bibliothek monoklonaler Antikörper, gewonnen aus Patienten mit:
- Autoimmun-Enzephalitis
- Autoimmunformen der Demenz
- anderen das zentrale Nervensystem beeinträchtigenden Autoimmunerkrankungen
- COVID-19
Wir sind laufend auf der Suche nach Lizenzpartnern oder Forschungskollaborateuren für:
- Forschungswerkzeuge
- Diagnostik
- Therapeutika
Darüber hinaus suchen wir Partner in der Industrie und im akademischen Bereich für die weitere Charakterisierung (z. B. Zielidentifizierung) unserer Antikörper.
Schlüsselpublikationen
Kreye J, Wenke NK, Chayka M et al. Human cerebrospinal fluid monoclonal N-methyl-D-aspartate receptor autoantibodies are sufficient for encephalitis pathogenesis. Brain 2016; 139:2641-2652.
doi: 10.1093/brain/aww208
Jurek B, Chayka M, Kreye J, et al. Human gestational N-methyl-d-aspartate receptor autoantibodies impair neonatal murine brain function. Ann Neurol 2019; 86:656-670.
doi: 10.1002/ana.25552
Kreye J, Reincke SM, Kornau H-C et al. A therapeutic non-self-reactive SARS-CoV-2 antibody protects from lung pathology in a COVID-19 hamster model. Cell 2020.
doi: 10.1016/j.cell.2020.09.049
Kreye J, Wright SK, van Casteren A et al. Encephalitis patient-derived monoclonal GABAA receptor antibodies cause epileptic seizures. J Exp Med 2021.
doi: 10.1084/jem.20210012
Reincke SM, Yuan M, Kornau H-C et al. SARS-CoV-2 Beta variant infection elicits potent lineage-specific and cross-reactive antibodies. Science 2022.
doi: 10.1126/science.abm5835
Management
BaoBab wird von einem erfahrenen und interdisziplinären Team von DZNE-Wissenschaftlern aus Berlin und Bonn gebildet, die sich der Translation von Grundlagenforschung in die klinische Anwendung verschrieben haben.
Unterstützt wird das Team von Experten der DZNE Technologietransferstelle und unserem Partner Ascenion.

Charitéplatz 1
intern: Bonhoefferweg 3


Scientific advisor und Head of BaoBab

intern: Bonhoefferweg 3
Scientific advisor

