ENABLE
Patient- and care-related benefits of amyloid PET imaging
Background and aims
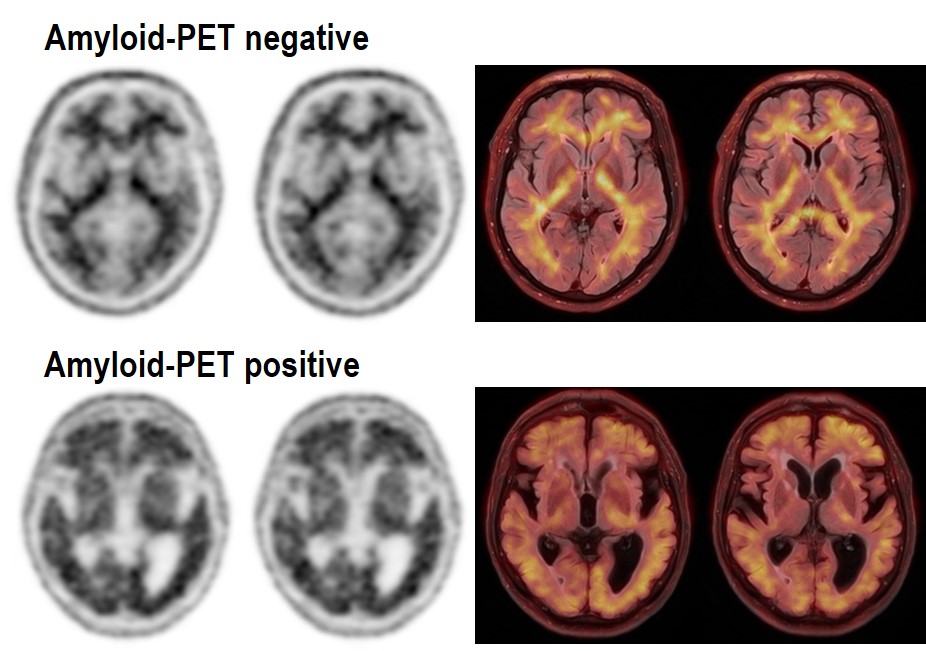
In the current care system, the majority of patients with dementia symptoms do not receive a diagnosis or receive a diagnosis of dementia of unknown cause. Even in patients who do receive a specific diagnosis of Alzheimer's disease dementia, in many cases this is associated with diagnostic uncertainty. Such diagnostic uncertainty interferes with the appropriate treatment, care, and counseling of dementia patients and their families. Amyloid PET examination is used to exclude or detect the presence of pathological protein accumulations in the brain that are typical of Alzheimer's disease. It can help to increase diagnostic certainty and thus improve the diagnosis and management of people with dementia. The aim of the ENABLE study is to test whether amyloid PET examination has the potential to improve treatment and care of people with dementia and thus preserve or slow down the loss of everyday functions in the German health care system.
The aim of a Coverage with Evidence Development (CED) study according o §137e of the German Social Code Book V (SGB V) is to determine whether a diagnostic or therapeutic procedure has a demonstrable (additional) benefit for patients under the conditions of the German health care system. The Federal Joint Committee (G-BA), the highest decision-making body of the joint self-administration in the German health care system, commissioned the DZNE and its partners on January 11, 2022, to test the benefit of an amyloid PET examination for patients with dementia of unclear cause or with an uncertain diagnosis of Alzheimer's disease according to the regulations of a CED study.
Overview
This is a two-arm, open-label, randomized, controlled, multicenter, superiority trial in a parallel-group design with blinded endpoint collection and analysis. In the study, 1,100 patients with mild to moderate dementia of unclear cause or with Alzheimer's dementia without sufficient diagnostic certainty after a specialist diagnosis will be included in a two-arm, open, randomized study. In one diagnostic arm, participants will receive an amyloid PET examination; in the other arm, such an examination will not be performed. Only patients in whom CSF diagnostics for amyloid detection could not be performed, were rejected or did not lead to a conclusive result will be included.
Course of the study
Participants will be examined over a 24-month period. The primary question of the effect of randomized amyloid PET diagnosis and associated improved diagnosis, treatment, and care on the ability to manage activities of daily living, as measured by the Amsterdam Instrumental Activities of Daily Living Questionnaire© (A-IADL-Q) score, will be assessed 18 months after randomization, followed by follow-up at an additional 6 months. Further questions are the effects on diagnostic confidence and treatment management of dementia patients.
Principle Investigator: Prof. Dr. Stefan Teipel
Start of the study: end of 2023
Status: multi centric, study under preparation
Study coordination / Project management
Dr. Annika Spottke
DZNE - Clinical Research Platform
annika.spottke(at)dzne.de