Areas of investigation/research focus
Neuronal Degeneration and Regeneration at Eye Level
Neurodegenerative diseases of the retina are a leading cause of vision loss with devastating consequences for the individual and our society. Inherited or acquired retinal dystrophies and age-dependent macula degenerations (AMD) are diverse groups of blinding diseases impacting many patients. It has been estimated that by 2040 over 280 million people will suffer from AMD worldwide, which might further increase in an aging society. Unfortunately, preventive or regenerative therapies effective against loss of retinal neurons and visual function are not yet available. Our aims are to decipher mechanisms of retinal degeneration and regeneration to control neuronal and glial disease responses as a unifying concept to prevent vision loss and facilitate regenerative therapies. Our main hypotheses are that common processes of retinal neurodegeneration, specifically inflammation, scar formation and tissue remodeling, might be a cause of vision loss, consequence of a failed regenerative program and source of novel therapeutic targets.
Our main research goals at the DZNE in Dresden are:
- To develop human (stem) cell-based model systems to study neuronal degeneration and regeneration in the retina.
- To decipher mechanisms linking retinal degeneration and regeneration – specifically those causing vision loss and limiting regeneration.
- To find new potential therapeutic strategies for retinal diseases.
Develop cell-based systems for retinal regenerative medicine.
- Our powerful mouse retina regeneration ex vivo assay recapitulates key processes of regeneration and failure thereof.
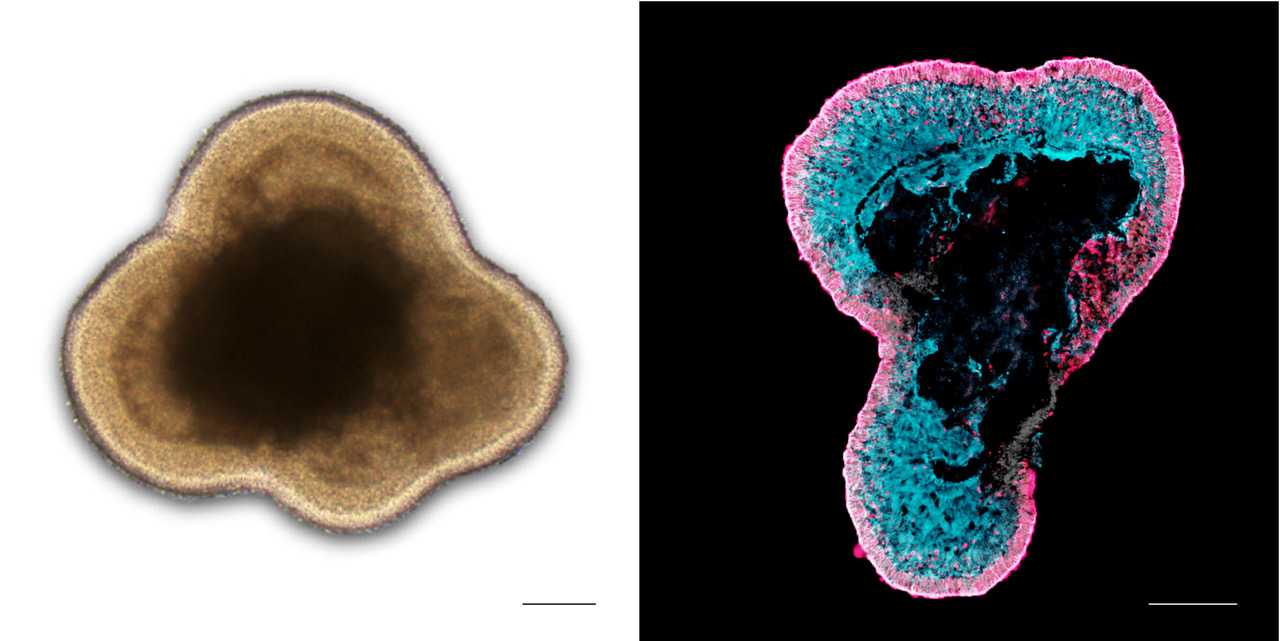
- Mouse and human pluripotent stem cell (hiPSC) derived organoid systems that recapitulate retinogenesis and facilitate disease modeling.
- Organoid models tuned for various studies: degeneration, regeneration, inflammation and cell-, drug- & gene-based therapies.
Decipher & overcome mechanisms limiting mammalian retinal regeneration.
- Stimulate glia derived neuronal regeneration: Our data showed cell death, stimuli and age–dependent regeneration, suggesting regulated restrictive mechanisms.
- Dissect mechanisms regulating and limiting regeneration using our retina ex vivo assay as a phenotypic testbed.
- Validate potential new regulators of regeneration in mice and human organoids.
Retinal disease modeling.
- Identify requirements for modeling inherited or acquired retinal dystrophies and age-related macula degeneration (AMD).
- Develop stem–cell based organoid models recapitulating retinal disease processes for applications in basic research and regenerative medicine.
- Determine pathomechanisms of common disease–associated processes, i.e. gliosis, tissue remodeling and scar formation, and neurodegeneration.
Please also visit: www.mokalab.org