Leibniz-Prize 2016

The neurons of the spinal cord lack the ability for self-repairment. If they are damaged, permanent paralysis or other health problems usually occur. Therefore, at the DZNE's Bonn site, Frank Bradke is researching how to release the brake that usually prevents the regeneration of cells of the central nervous system. The neurobiologist hopes that his studies will open up new avenues for the treatment of spinal cord injuries in the long term. In March 2016, Bradke received the Leibniz Prize (worth 2.5 million euros) for his groundbreaking work.
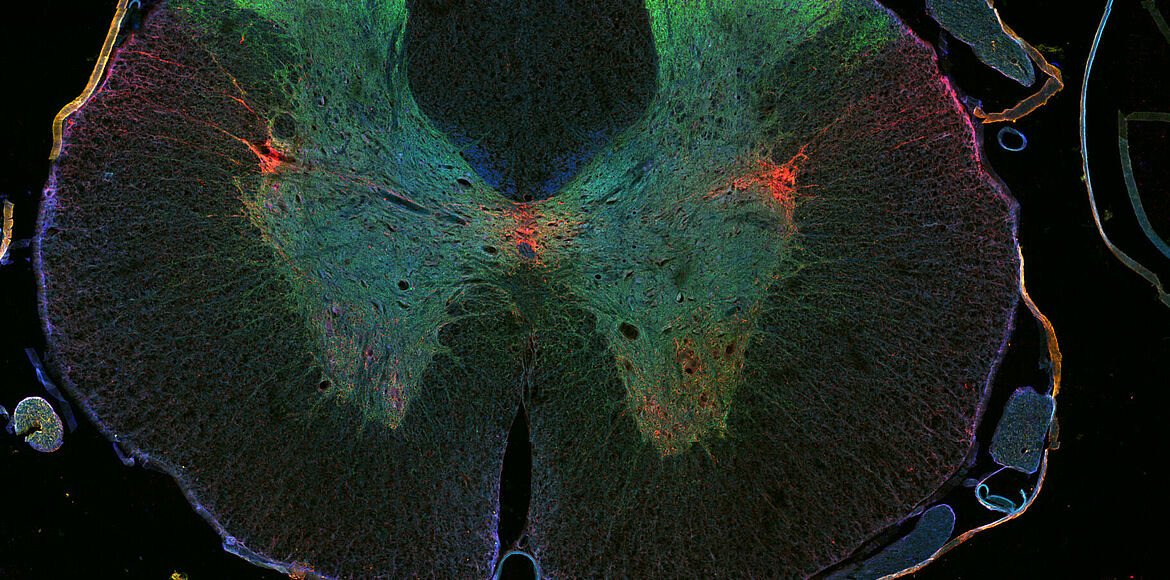
Nerve fibers meander through the human body in a way similar to the wiring of a computer network. Control signals thus run from the brain to the tips of the toes, while sensory information flows back the opposite way. The bowel, heart and other organs are also connected to this network of neurons. This wide reaching web ensures that we can move, perceive our environment and it regulates (often unconsciously) almost every function of the human body. As with a sequence of relay stations, electrical impulses are thereby relayed from neuron to neuron. Damage to these connections can have serious consequences. The brain and the muscles may be intact, but if the network in between them is disturbed, the affected muscles can no longer be subject to targeted control. They may even remain paralyzed.
A question of location
Whether injured neurons recover, depends essentially on their location. In the limbs and torso they have the talent for self-healing. Although damaged nerve cells are not replaced by new ones, they can regrow and thus make new connections with other neurons. Therefore, even a severed and reattached hand can regain some degree of sensitivity and mobility. However, the cells of the central nervous system – that is the brain and spinal cord - lack this ability to regenerate. Why? And, what causes neurons to grow and how could this be supported?
Long connections
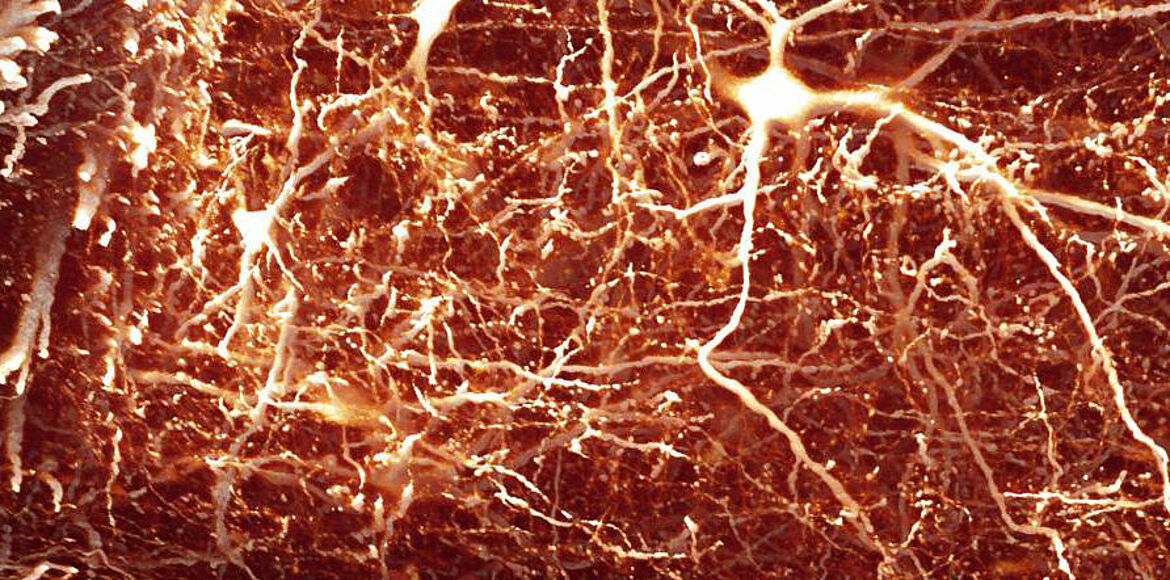
For around 25 years, Frank Bradke has been working on these questions. He is particularly interested in the “axons”, the projections through which nerve cells stay in contact with each other. In the narrow space of the brain, they only have to bridge relatively short distances. Things are different in the spinal cord: here, some axons extend to more than a meter. If these connections are damaged or even severed, cellular communication is impaired.
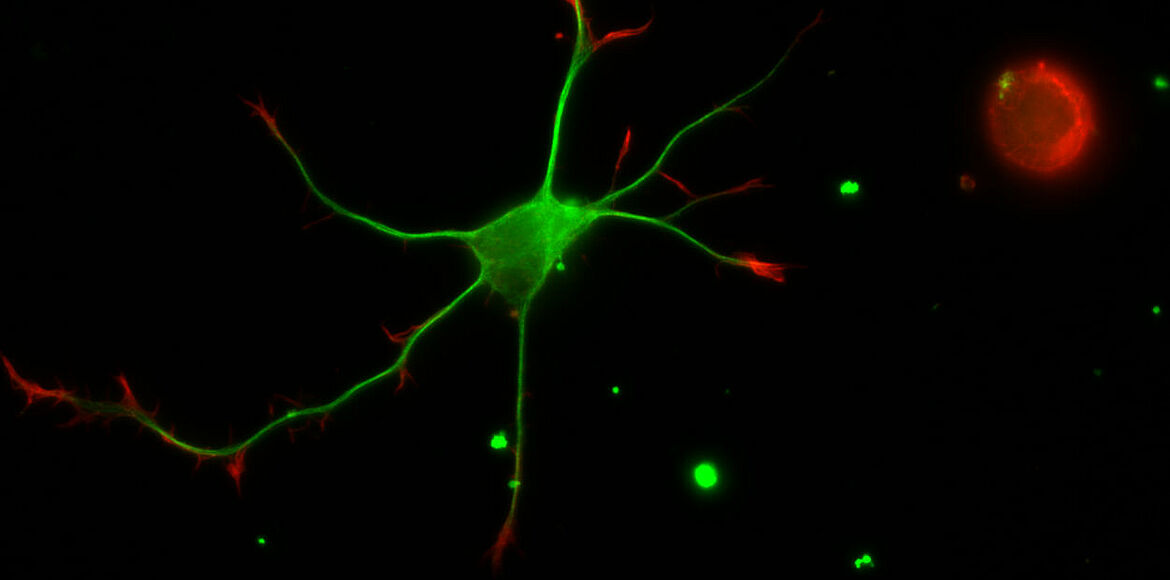
Bradke and colleagues found out that so-called microtubules play a key role in the growth of axons. These tubular protein filaments are part of the cellular skeleton that provides the axons with shape and stability. At the same time they serve as transport routes for neurochemicals.
Induced growth boost
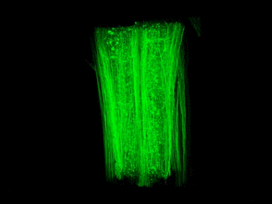
In laboratory studies, the researchers succeeded in proving that certain cancer drugs stimulate the microtubules to grow. As a result, the axons reach out again. Moreover, the scientists found that these drugs reduce scarring of the surrounding tissue. This additionally promotes regeneration of neurons. In fact, when rats with spinal cord injuries were treated accordingly, their mobility significantly improved.
“Our approach is to turn the nerve cell into a daring driver, who runs over every stop sign”, is how Bradke describes his team’s research strategy. “These stop signs include, for example, chemicals that are released by the scar tissue surrounding the neuron. Besides, there are also mechanisms in the cell that prevent growth. Getting rid of all these stop signs is a difficult task. That is why we want to understand what drives the growth of axons. We would like to boost up this engine, so to speak, and release the brake on growth.”
Long way to therapy
However, it may take years for patients to benefit from this research. “Gradually, we are understanding better and better which mechanisms are crucial for the regeneration of neurons. An ideal treatment for spinal cord injury should stimulate microtubules to grow and reduce scarring. However, our picture of these processes is far from complete. Therefore, clinical trials or even therapy are not yet within reach. There is still a long way to go”, says Bradke.