Neurons Able to “Talk” Cannot Regenerate
Adult neurons of the central nervous system do not regenerate if they are damaged, for example following a spinal cord injury. A study by DZNE researchers published in the scientific journal “Neuron” provides evidence that this inability for recovery is closely related to the neuron’s capacity to communicate with each other. Specifically, the scientists found that two proteins which are essential for so-called synaptic transmission between neurons prevent growth of cell processes. These findings shed light on the mechanisms of neuronal injury and might help in the search for better treatments.
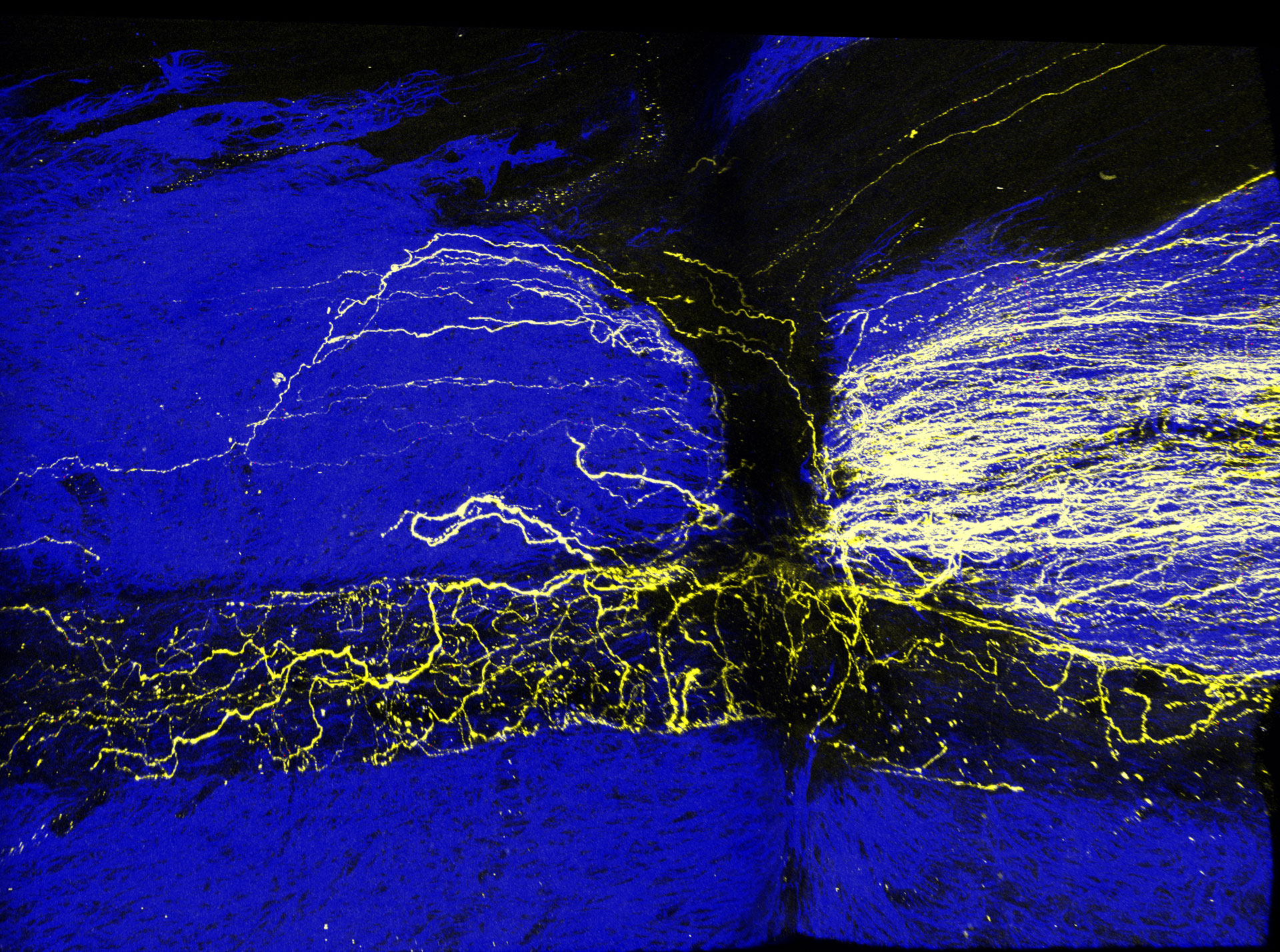
During embryonic development, neurons grow processes called “axons”, by which they reach out to other neurons and ultimately make connections to them. But this ability is subsequently lost. In fact, neurons of the adult brain and the spinal cord are unable to regenerate and form new axons. If, for example, the spine is harmed, this can have serious consequences, as the nerve fibers running in the spinal cord do not heal or regrow on their own. As a result, communication between the brain and the rest of the body can be disrupted permanently. This may lead to paralysis and other disabilities. At the DZNE’s Bonn site, a team headed by Prof. Frank Bradke is exploring the molecular mechanisms underlying such injuries. “Adult neurons do not regenerate after damage. On the other hand, adult neurons are characterized by being networked with others and exchanging signals with them. This suggests that they cannot do both at the same time. In other words, these cells can either grow or talk to each other. Our findings support this hypothesis”, Bradke says.
Genetic and Lab Studies
For the current study, the researchers focused on the dorsal root ganglion (DRG), a neuronal structure in the back area that mediates signal exchange between the spinal cord and the rest of the body. “DRG neurons are special because under certain conditions they acquire competence to regrow axons and are thus able to regenerate. Studying these neurons may therefore provide insights into the mechanisms of regeneration”, says Brett Hilton, a postdoc researcher in Bradke’s group and first author of the current publication in “Neuron”. “We hypothesized that gene expression in these neurons may provide insights into what underlies the ability to grow axons. Therefore, we examined which genes are up- or down-regulated – on the one hand during embryonic development and on the other hand when adult DRG neurons acquire growth capacity. Through these analyses, combined with studies in cell cultures and mice, we eventually came across two proteins. These proteins are found in the synaptic terminals of neurons and prevent adult neurons from developing axons. In other words, they suppress regeneration.”
The identified molecules named “Munc13” and “RIMs” are present in the so-called presynaptic terminal, the outermost end of an axon. Both proteins are involved in a process referred to as “vesicle priming” that ultimately leads to the release of neurotransmitters, chemical compounds through which neurons communicate with each other. “Experiments, in which we were able to activate and deactivate these proteins, so to speak, showed that Munc13 and RIMs inhibit regeneration of neurons”, Hilton says. “We don’t know the mechanisms by which this happens. But obviously these proteins play an important role.”
Tests with the Drug “Baclofen”
In view of the significance of synaptic transmission, the researchers also tested whether attenuating this transmission promoted axon growth. To this end, they administered the drug Baclofen to mice with spinal cord lesion. Baclofen reduces neuronal excitability and synaptic transmission. Therefore, the drug is used in humans when neurons fire uncontrollably, for example, in cases of spasticity or spinal cord injury. Analyses of neurons from these mice revealed that treatment with Baclofen indeed stimulated axon growth and regeneration in the injured spinal cord. “Data from other studies show that Baclofen promotes motor capability in individuals with spinal cord injury. This might relate to the effect of Baclofen on muscle spasms and undamaged neurons. However, our findings suggest that Baclofen might enhance the regeneration of damaged neurons”, Bradke says. “Overall, our data indicate that mechanisms involving synaptic transmission, and in particular the presynaptic terminal, are preventing neuronal regeneration. However, it takes even more than regrowing axons to alleviate the effects of spinal cord injury. The consolidation of new synaptic circuits is equally important and requires the development of novel rehabilitation therapies.”
Original publication
An active vesicle priming machinery suppresses axon regeneration upon adult CNS injury.
Brett J. Hilton et al.
Neuron (2021).
DOI: 10.1016/j.neuron.2021.10.007