Areas of investigation/research focus
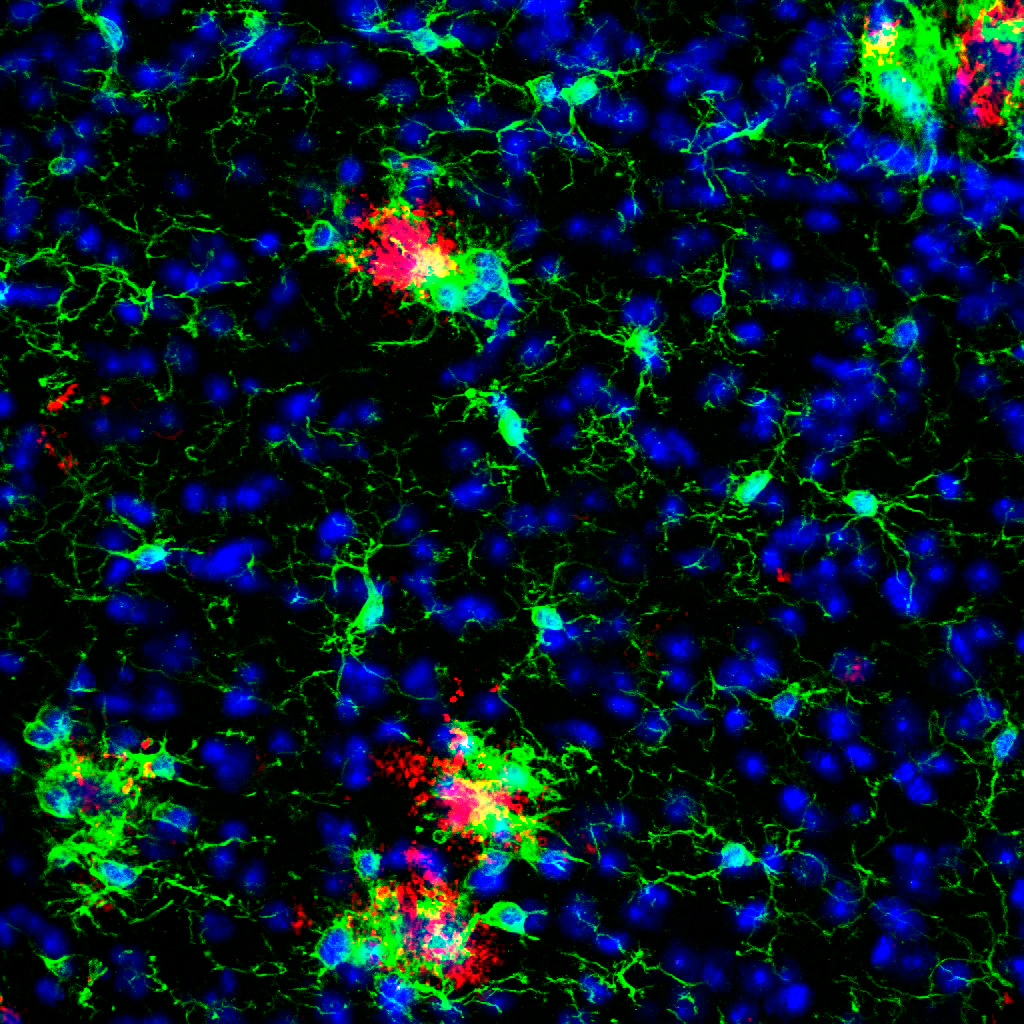
Our research focuses on microglial dysfunction and alterations in neuronal and glial signaling contributing to the pathogenesis of neurodegenerative disorders like Alzheimer´s disease (AD), Niemann-Pick type C (NPC) and Frontotemporal lobar degeneration (FTLD). Better understanding of cell-specific pathological changes and cross-talk between different brain cells will help us to reveal novel therapeutic targets for medical intervention.
Microglial activation is one of the key pathological hallmarks of neurodegeneration. It is proposed that in AD microglia are inefficient in removing ß-amyloid deposits. To study microglial contribution to amyloid plaque burden, we designed a novel ex vivo model (brains in a dish) where we culture together organotypic brain slices from aged, amyloid-bearing and young wild-type mice. The co-culture paradigm or only the exposure of old AD microglia to soluble factors produced by young microglia was sufficient to “rejuvenate” their phagocytic capacity and trigger amyloid plaque clearance. Our goal is to identify compounds aimed to instruct AD microglia to clear amyloid and understand how microglial immunomodulation can be used to mitigate neuropathology. A future challenge will be to therapeutically enhance beneficial responses of microglia -such as phagocytosis- to ameliorate AD.
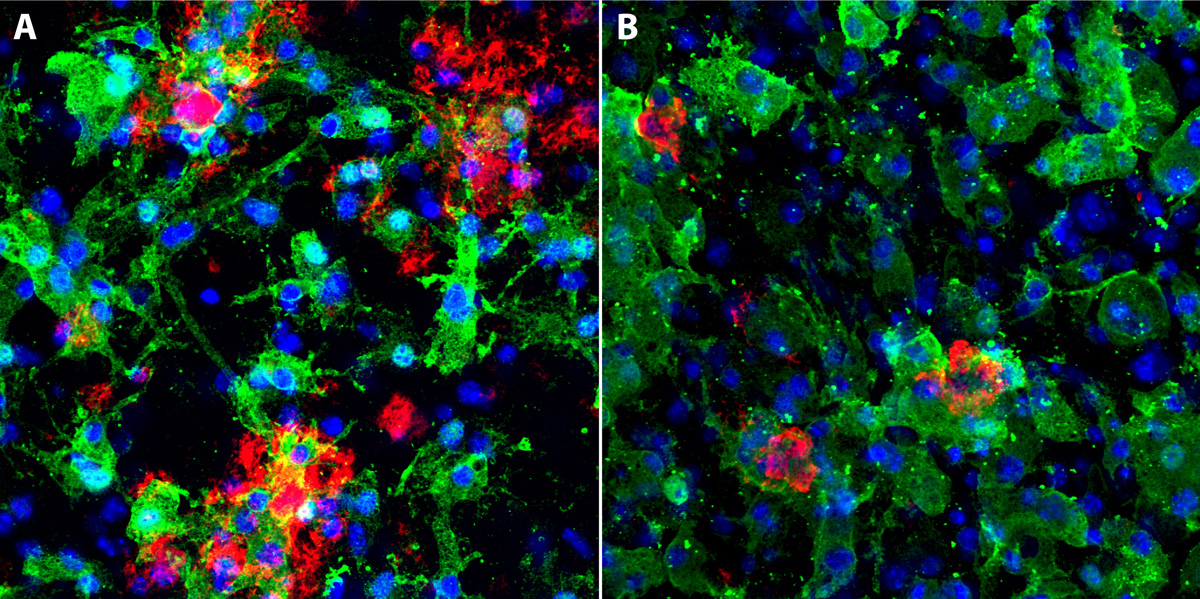
Beyond AD, microglial contribution becomes appreciated in other neurodegenerative conditions such as FTLD and NPC. Those diseases are of particular interest for our research due to common mechanisms shared between lysosomal storage disorders and neurodegeneration. NPC is a lysosomal storage disorder that is due to many shared pathological features also called juvenile AD as it is mainly affecting children and young adults. Our research goal is to characterize microglial dysfunction and dissect common and disease-specific molecular alterations in AD, FTLD and NPC.
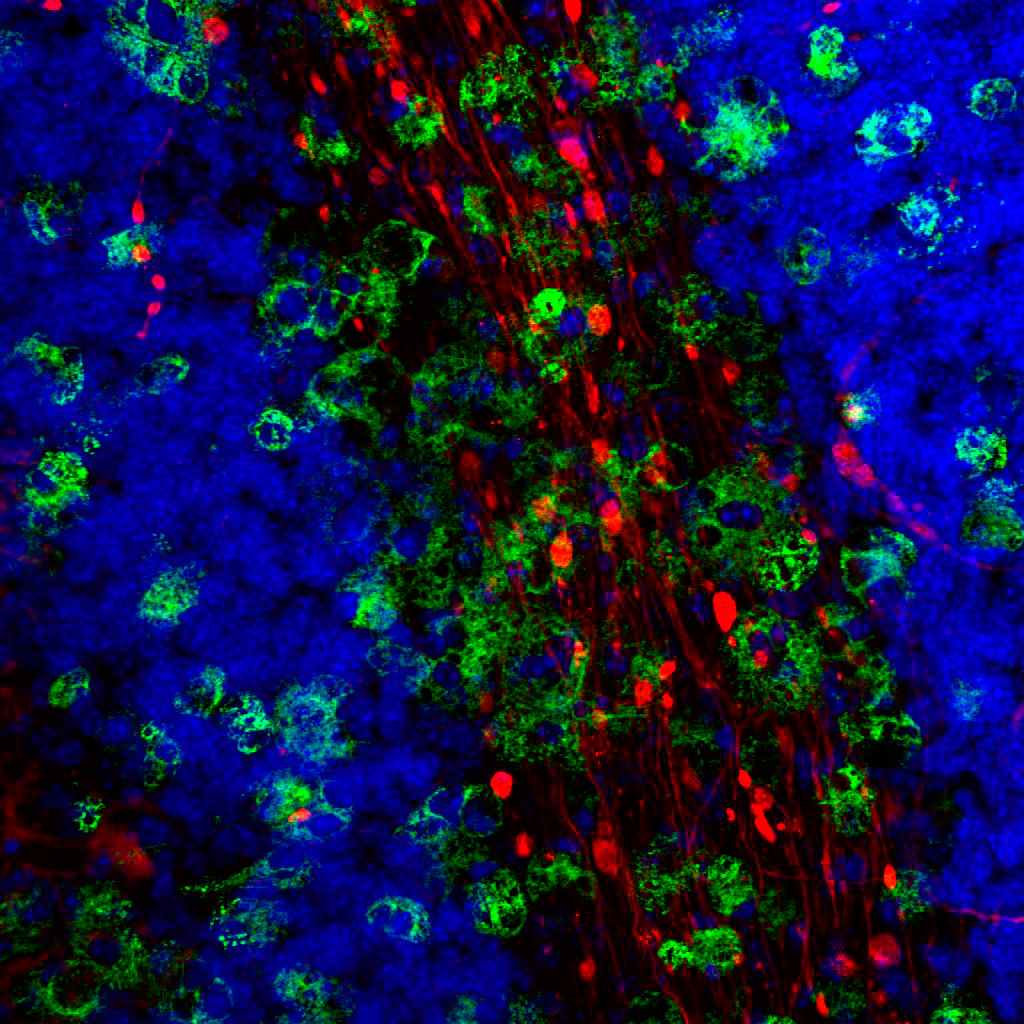
We combine our expertise in isolation and characterization of primary cells (neurons, astrocytes, microglia and organotypic brain slices) with in vivo approaches (transgenic mouse models of neurodegeneration) to elucidate complex pathological changes of different brain cells. Using cell biological, biochemical, immunohistochemical and proteomic analysis we asses the contribution of cell-type-specific alterations to disease onset and progression with the final aim to identify potential drug targets for treatment of those devastating neurodegenerative disorders.

