Areas of investigation/research focus
My group is interested in defining key molecular pathways in the pathogenesis of neurodegeneration with a goal of identifying specific targets for therapeutic intervention. In particular, we focus on the second most common neurodegenerative disorder Parkinson’s disease (PD) and more rare neurological disorders such as those in the “Neurodegeneration with Brain Iron Accumulation” (NBIA) family.
Our laboratory utilizes induced pluripotent stem cell (iPSC) technology to establish human disease models of relevant central nervous system cell types such as vulnerable dopaminergic neurons and glia from patient’s somatic cells. Our overall objective is to use these model systems to unravel cell autonomous and non-cell autonomous mechanisms leading to the preferential demise of specific neuronal subsets in PD and related disorders.
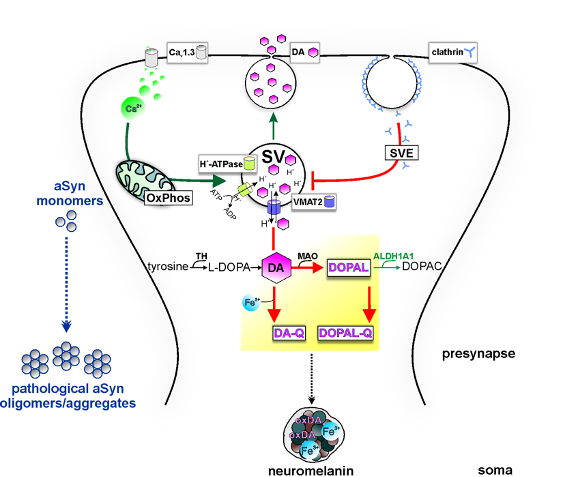
In PD the most susceptible cells are the dopaminergic neurons in the substantia nigra, a structure located in the midbrain. However, the mechanisms underlying their preferential demise are still under investigation. Mitochondrial malfunctions and oxidative stress are considered some of the major causes of neurodegenerative processes. In addition, abnormal metabolism of the neurotransmitter dopamine (DA) is suggested to play a critical role in pathology through excess generation of toxic species (e. g. DA quinones, DOPAL) and oxidative stress.
In 2017, during my postdoctoral fellowship at Northwestern University, Chicago, we discovered a human-specific and time-dependent pathological cascade of mitochondrial and lysosomal dysfunction mediated by oxidized DA in long-term cultures of iPSC-derived midbrain dopaminergic neurons from genetic and sporadic PD patients (Burbulla et al., Science, 2017). Interestingly, this pathogenic cascade was not found in PD mouse models due to species-specific differences in DA metabolism, which could partially explain the incomplete recapitulation of human pathology in currently available animal models. Oxidized DA derivates were shown to modify specific lysosomal enzymes in iPSC-derived patient neurons, resulting in reduced lysosomal enzyme activity and accumulation of alpha-synuclein, a key feature in brains of PD patients. In follow-up translational studies we were able to increase the reduced lysosomal enzyme activity using small molecule modulators or drug repurposing strategies of FDA-approved drugs and thus diminish the accumulation of oxidized DA and alpha-synuclein (Burbulla et al., Science Translational Medicine, 2019; Burbulla et al., JCI Insight, 2021). These results emphasize the value of human disease models to identify relevant targets for development of new treatments in PD.
Interestingly, PD shares pathological hallmarks including brain iron accumulation, elevated accumulation of the polymer neuromelanin – a major iron chelator in dopaminergic neurons - and degeneration of midbrain DA neurons with the rare, neurological disorder β-propeller-associated neurodegeneration (BPAN), a subtype of NBIA disorders. We were the first to unravel mitochondrial and lysosomal alterations, and increased iron levels in a patient-derived neuronal model of BPAN (Seibler*, Burbulla* et al., Brain, 2018). Similar to research of PD pathology, studies using human models of disease-relevant cell types are needed for a better understanding of the disease course and the development of therapeutics for BPAN and other forms of NBIA.
Combining genomic approaches with functional physiological assays, our group evaluates progression of disease pathology by studying key pathogenic mechanisms in long-term cultures of vulnerable midbrain dopaminergic neurons generated from patient iPSCs focusing on the imbalance of DA metabolism and emergence of toxic DA by-products through mitochondrial dysfunction, altered enzymatic DA degradation, and iron dyshomeostasis. As preferential vulnerability of midbrain dopaminergic neurons in PD and subtypes of NBIA may not only arise from cell autonomous mechanisms, our research also explores neuron-glia crosstalk to investigate the contribution of non-cell autonomous mechanisms to neurodegeneration.
A better understanding of disease progression and identification of relevant targets for the development of new treatments will be important for a large cohort of patients affected by neurodegenerative diseases.